In some of the simulations that we discussed in a previous blog post on molecular dynamics (MD) studies on anion exclusion, chloride never enters the montmorillonite interlayers. From such results, authors have argued for complete anion exclusion from interlayers, and thereby supported ideas of a multi-porous structure of compacted water saturated bentonite. It is, however, glaringly obvious that these simulations are not even close to being converged, and that they should not have been published in the first place. It is also clear that chloride do enter interlayers in properly conducted MD studies, in both tri- and bi-hydrated sodium montmorillonite.
Reasonably, it should only be a matter of time before researchers that support ideas of complete exclusion manage to perform MD simulations that better reflect anion equilibrium in montmorillonite. As such possible future simulations will confirm that anions have access to interlayers,1 I have in the back of my mind wondered about potential consequences. Will earlier publications be retracted? Will the entire “mainstream view” of the structure of compacted bentonite fall into oblivion? (I wish.) From this perspective I find it a bit amusing that no further MD simulation has been published to support complete anion exclusion for the last ten years (as far as I’m aware).
Hsiao and Hedström (2022)
Here, I want to highlight a considerably more recent MD study that actually has advanced our understanding of chloride equilibrium in montmorillonite: “Salt Effect on Donnan Equilibrium in Montmorillonite Demonstrated with Molecular Dynamics Simulations” (Hsiao and Hedström, 2022). This is a straightforward continuation of the study by Hsiao and Hedström (2015), which we discussed in detail in the earlier MD blog post. I will refer to these two studies as Hsi22 and Hsi15, respectively.
Hsi15 simulated bi-hydrated sodium montmorillonite interlayers in contact with a bulk compartment with two different NaCl concentrations (1.67 M and 0.55 M), and showed that these systems obey the rules for Donnan equilibrium, albeit with a substantial non-electrostatic contribution to the free energy (non-ideal conditions). Hsi22 continue this work by presenting a complementary simulation at a third NaCl concentration (1.0 M), and by performing corresponding simulations for CaCl2, at bulk concentrations 0.14 M, 0.28 M, 0.52 M, and 0.84 M (with all interlayer cations then being calcium, obviously). With chloride equilibrium simulations for several background concentrations for both Na- and Ca-montmorillonite, but for otherwise identical systems, Hsi22 are able to make thorough comparisons with Donnan equilibrium theory.
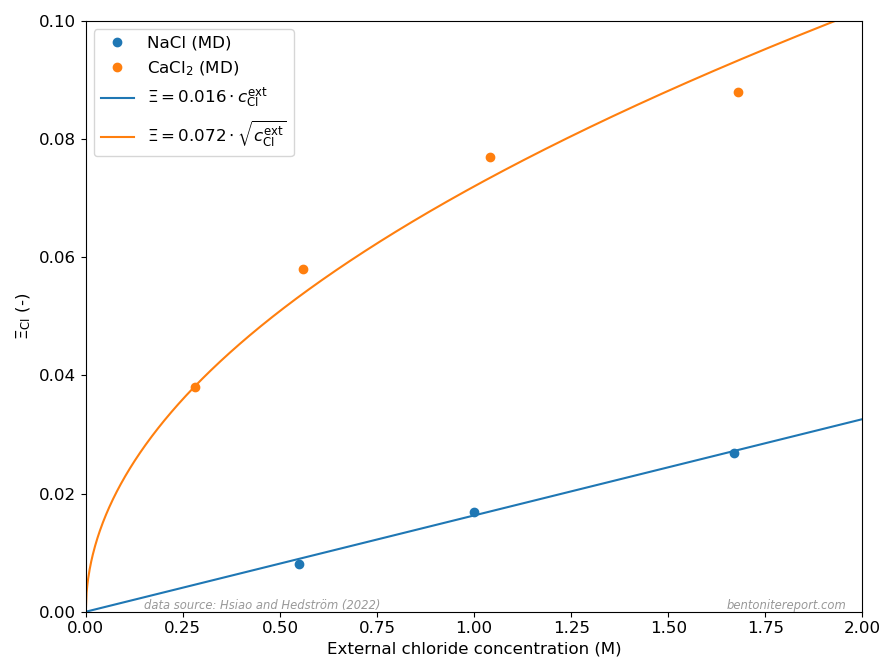
Chloride equilibrium — just as any other ion equilibrium — is conveniently expressed via the ratio \(\bar{\mathrm{c}} / c^\mathrm{ext}\), where \(\bar{\mathrm{c}}\) is the clay concentration and \(c^\mathrm{ext}\) is the corresponding bulk concentration. In a Donnan equilibrium context this concentration ratio may be identified with the ion equilibriumcoefficient2 \begin{equation}
\Xi_\mathrm{Cl} \equiv \frac{c^\mathrm{int}_ \mathrm{Cl}}
{c^\mathrm{ext}_\mathrm{Cl}}
\end{equation} where \(c^\mathrm{int}_\mathrm{Cl}\) is the interlayer concentration of chloride in a homogeneous bentonite domain in equilibrium with an external solution with chloride concentration \(c^\mathrm{ext}_\mathrm{Cl}\).
For a 1:1 system (e.g. NaCl in contact with Na-montmorillonite) a good approximation for \(\Xi_\mathrm{Cl}\) at low external concentration is3 \begin{equation} \Xi^{1:1}_\mathrm{Cl} \approx \Gamma^2 \frac{c^\mathrm{ext}_\mathrm{Cl}}{c_\mathrm{IL}} \tag{1} \end{equation} where \(c_\mathrm{IL}\) is the structural montmorillonite charge expressed as a monovalent interlayer concentration (in the model of Hsi22 and Hsi15, \(c_\mathrm{IL} = 4.23\) M) and \(\Gamma\) is a mean activity coefficient ratio for NaCl (more on that below).
While the ion equilibrium coefficient in eq. 1 depends linearly on the external concentration, the corresponding quantity for a 2:1 system (e.g. CaCl2 in contact with Ca-montmorillonite) depends on the square-root of the external concentration (note that the Cl concentration in a CaCl2 solution is twice that of CaCl2) \begin{equation} \Xi^{2:1}_\mathrm{Cl} \approx \Gamma^{3/2} \sqrt{\frac{c^\mathrm{ext}_\mathrm{Cl}}{c_\mathrm{IL}}} \tag{2} \end{equation} where \(\Gamma\) here is to be understood as a different mean salt activity coefficient ratio (for CaCl2).
The different dependencies on \(c^\mathrm{ext}_\mathrm{Cl}\) for Na- and Ca-systems, expressed in eqs. 1 and 2, are clearly reproduced in the MD results presented in Hsi22. as shown here
The dots show the chloride equilibrium coefficients as calculated in Hsi22 and Hsi15, primarily from evaluated potentials of mean force evaluated using the adaptive biasing force method. The corresponding curves in the above diagram are my attempt at fitting eqs. 1 and 2 to these MD results.
It should be noted that the linear and square-root dependencies of eqs. 1 and 2, respectively, presume that the activity coefficient ratios (\(\Gamma\)) are essentially independent of \(c_\mathrm{Cl}^\mathrm{ext}\). The successful fits of eqs. 1 and 2 thus demonstrates that this is the case for the MD equilibrium coefficients.4 Hsi22 make a deeper analysis and show that the specific values of the activity coefficient ratios correspond to differences in excess chemical potential for the salt of 1.35 kT and 1.25 kT, respectively, for the Na- and Ca-systems. Such values reflect a quite profound non-ideal behavior, which may be related to the details of the simulations (e.g. non-polarizable force fields) rather than corresponding to an actual excess barrier.
The main message in Hsi22 is nevertheless clear: Results from MD simulations of chloride in Na- and Ca-montmorillonite are consistent with Donnan equilibrium theory. This means, in particular
- \(\Xi_\mathrm{Cl}\) is linear for NaCl and has a square-root dependence for CaCl2
- For a given external chloride concentration and density, the amount chloride entering the interlayers is much larger in Ca-montmorillonite as compared to Na-montmorillonite
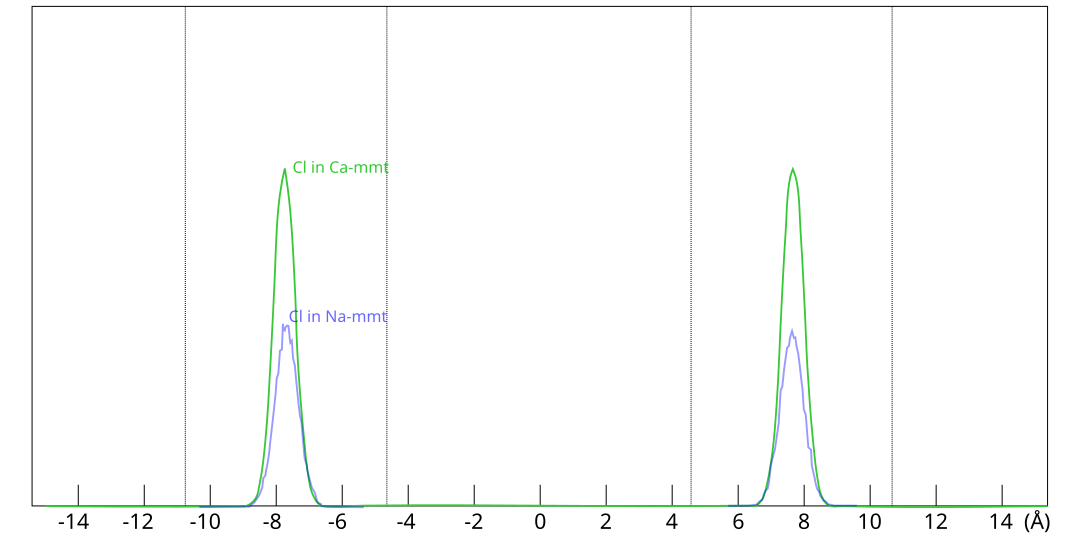
To be clear, the much larger amount of chloride predicted to be found in Ca-montmorillonite has nothing to do with any notions of different “anion-accessible” pore spaces, but is a direct consequence of Donnan equilibrium. In these simulations, all chloride is located at the exact same place within the clay, as shown here
This figure shows evaluated chloride density profiles in the direction perpendicular to the mineral layers in MD simulations of Na-montmorillonite (Hsi15) and Ca-montmorillonite (presented in the supporting information to Hsi22). While I have arbitrarily scaled the profiles along the y-axis in the above figure for visualization purposes, emphasis is here on the identical position within each interlayer. Note that the simulated system contains two separate interlayers, indicated by dotted vertical lines.5
One lesson from these results is that researchers who struggle with getting chloride to enter interlayers in their simulations could use CaCl2 rather than NaCl. At e.g. an external chloride concentration of \(\sim\)0.5 M, the amount chloride in the clay is about seven times larger in Ca- as compared with Na-montmorillonite, which substantially reduces the required convergence time for the simulation.
These results also highlight the urgent need for empirical data. As I pleaded for when concluding the assessment of chloride equilibrium concentrations in Na-bentonite, labs all over the place should routinely produce and publish ion equilibrium measurements. It is certainly a failure of the bentonite research field that no published empirical data exists that can be used to compare with these theoretical results. Indeed, as far as I’m aware, no published systematic empirical data exists at all, for anion equilibrium concentrations in calcium dominated bentonite.6
Note that the implication of the results discussed here is not simply some noted interesting difference in chloride equilibrium in different types of montmorillonite. Rather, as the results indicate that montmorillonite interlayers play by the rules of ordinary Donnan equilibrium, they are an additional blow to the entire contemporary multi-porous model description of compacted water saturated bentonite.
Footnotes
[1] As I often nag about on this blog, it is quite silly to use complete anion exclusion as a starting point when studying compacted bentonite, and then trying to “confirm” such a notion with e.g. MD simulations. There is no rationale for this assumption in the first place; as we have discussed earlier, the idea seems to have originated from misunderstanding the Poisson-Boltzmann equation. Moreover, there is solid empirical evidence for salt entering interlayers, in particular from measured swelling pressure response.
[2] Hsiao and Hedström (2022) call this ratio a partition coefficient, which complies with the scientific literature on e.g. polymer membranes. As I discussed here, I have chosen to stick with some of my own terminology. I hope this does not cause unnecessary confusion.
[3] For details on these approximations, see e.g. Birgersson (2017)
[4] That the activity coefficient ratios do not depend strongly on external concentration in this concentration interval is also compatible with the mean salt approach that I have suggested to use for compacted bentonite. For the external solutions, mean salt activities varies quite little in this concentration range, and since the interlayer concentrations only varies with a few percent, it make sense to assume that the interlayer activities basically remain constant. Hsiao and Hedström (2022) actually note that the undulation pattern in the potential of mean force in the direction of the reaction coordinate is essentially independent of the external solution, and conclude that the interlayer environment is essentially independent of external conditions.
[5] The nearly identical profiles within each interlayer is also a confirmation that these simulations are properly converged.
[6] An indication that CaCl2 in Ca-montmorillonite behaves as discussed is found here.